Commencement of Pivotal Validation Program for FDA 510(k) Submission: First Participant Enrolled
BlinkLab announced the commencement of its validation program supporting 510(k) submission, with the first child participant now enrolled and having completed initial testing using BlinkLab Dx1.

Highlights:
- First Participant Enrolled in FDA 510(k) Pivotal Validation: BlinkLab has enrolled and successfully completed testing of its first participant using BlinkLab Dx1, marking the commencement of its Pivotal Validation for its autism diagnostic aid.
- Study Execution Led by CRO Partner: The study is led and conducted by BlinkLab’s Contract Research Organisation, IQVIA-MCRA, to ensure full adherence to study protocols, data quality standards, and regulatory requirements.
- Builds on Strong Pilot Study Performance: The commencement of the study follows a successful pilot study (n=485), where BlinkLab Dx1 demonstrated 83.7% sensitivity and 84.7% specificity, significantly exceeding target performance thresholds of >65% for both.
- Clear Path Toward FDA Submission: Recruitment is expected to complete within ~8 months, with 510(k) FDA submission targeted by year-end 2026.
BlinkLab Limited (ASX:BB1) (“BlinkLab” or the “Company”) is pleased to announce the commencement of its validation program supporting 510(k) submission, with the first child participant now enrolled and having completed initial testing using BlinkLab Dx1. The validation program is expected to generate the pivotal core clinical and usability evidence required to support BlinkLab’s planned FDA 510(k) submission. This validation program follows the successful completion of the pilot study, with results announced in October 2025¹.
FDA 510(k): Usability and Clinical Validation Program
BlinkLab has initiated its FDA-enabling validation program for its pediatric autism diagnostic aid, marking a major milestone with the enrollment of its first participant. In alignment with FDA expectations² and in collaboration with global CRO IQVIA, the study begins with a dedicated usability phase. This phase is designed to ensure that clinicians and caregivers can safely and effectively use the BlinkLab platform in real-world settings, including correct operation of the system and appropriate interpretation of outputs. Consistent with regulatory guidance, this usability phase will seamlessly transition into the clinical validation phase without further announcements. The study is designed to evaluate diagnostic performance against established standard-of-care assessments for autism, including key measures such as sensitivity, specificity, and predictive values (NPV and PPV). Participant recruitment for the validation program is expected to take approximately 8 months.
BlinkLab is on track to complete these activities and submit its regulatory package to the U.S. Food and Drug Administration by the end of 2026.
BlinkLab’s device is intended for use as an aid in diagnosis and is designed to support, not replace, clinical decision-making. By combining objective digital biomarkers derived from patient behaviour with clinician expertise, BlinkLab aims to enable earlier, more accurate, and more accessible and scalable autism assessment.
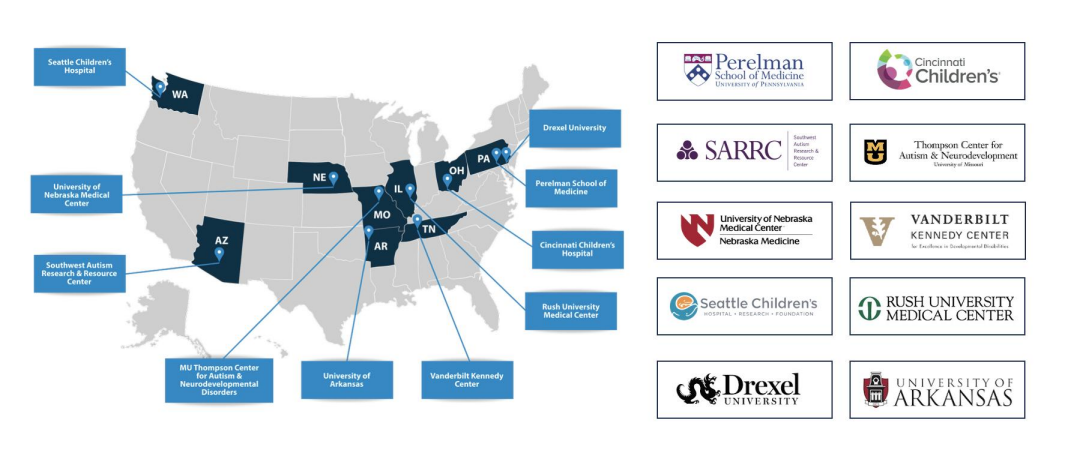
FDA 510(k) Pivotal Study: Centers Participating
The registrational trial for BlinkLab Dx1 is using a double-blinded, multi-center, within-subjects comparison study design to compare the technology with clinician’s professional assessments of children using gold standard methods as a reference. The main study phase of the trial is taking place across a network of 10 leading U.S. clinical sites, including Cincinnati Children’s Hospital³, Seattle Children’s Hospital⁴, University of Pennsylvania⁵, MU Thompson Center for Autism & Neurodevelopmental Disorders⁶, Southwest Autism Research & Resource Center⁷, University of Nebraska Medical Center⁸, Vanderbilt Kennedy Center⁹, Rush University Medical Center¹⁰, Drexel University¹¹, and University of Arkansas¹². Together, these sites represent a geographically diverse and demographically representative sample of the U.S. population, each with a proven track record in autism research and clinical trial execution.
BlinkLab CEO and Managing Director, Dr Henk-Jan Boele, commented:
“We are excited to initiate this next phase of our FDA 510(k) regulatory program. It marks what I believe is the most important milestone for BlinkLab to date, following years of focused development, extensive testing, optimisation of our tech, close collaboration and meetings with the FDA, and securing IRB approvals to support clinical execution. With a clear pathway toward FDA submission and strong execution underway, we believe BlinkLab is well positioned to transform how autism is assessed in clinical practice.”
This announcement has been authorised for release by the Board of BlinkLab Limited.
References
- ASX Announcement (22 October 2025) – “Pilot Study Confirms High Diagnostic Accuracy and Readiness for FDA Trial”
- https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-882/subpart-B/section-882.1491
- ASX Announcement (26 August 2025) – “Two additional strategic partnerships with top ranked U.S. institutions advance BlinkLab’s FDA submission”
- ASX Announcement (26 August 2025) – “Two additional strategic partnerships with top ranked U.S. institutions advance BlinkLab’s FDA submission”
- ASX Announcement (21 August 2025) – “BlinkLab Onboards University of Pennsylvania Perelman School of Medicine as Third Clinical Site for its FDA 510(k) Diagnostic Trial"
- ASX Announcement (17 September 2025) – “BlinkLab Engages The University of Missouri’s Thompson Center for Autism & Neurodevelopment as its sixth Clinical Site for FDA510(k) Diagnostic Trial”
- ASX Announcement (22 May 2025) – “BlinkLab Engages First Clinical Site for Main Study Phase of FDA 510(k) Diagnostic Trial"
- ASX Announcement (8 July 2025) – “BlinkLab Expands U.S. Clinical Trial Network: University of Nebraska Medical Center Engaged as Second Site in Main Phase of FDA 510(k)Study”
- ASX Announcement (18 September 2025) – “BlinkLab Onboards ‘The Vanderbilt Kennedy Center’ as Seventh Clinical Site for FDA 510(k) Autism Trial”
- ASX Announcement (13 November 2025) – “Rush University Medical Center Joins BlinkLab’s Pivotal U.S. Autism Diagnostic Trial”
- ASX Announcement (12 February 2026) – “BlinkLab Expands Pivotal FDA Trial Network to Nine Elite U.S. Sites Ahead of Imminent Study Commencement”
- ASX Announcement (16 March 2026) – “BlinkLab Completes Clinical Trial Network for Pivotal FDA Study with addition of Tenth Clinical Site”
Invest in BlinkLab
Interested in joining BlinkLab’s growth journey? Explore our Investor Centre.


